FILTER INTEGRITY ANALYZER
- Engineered in New York
- Made in India for Pharma Industries
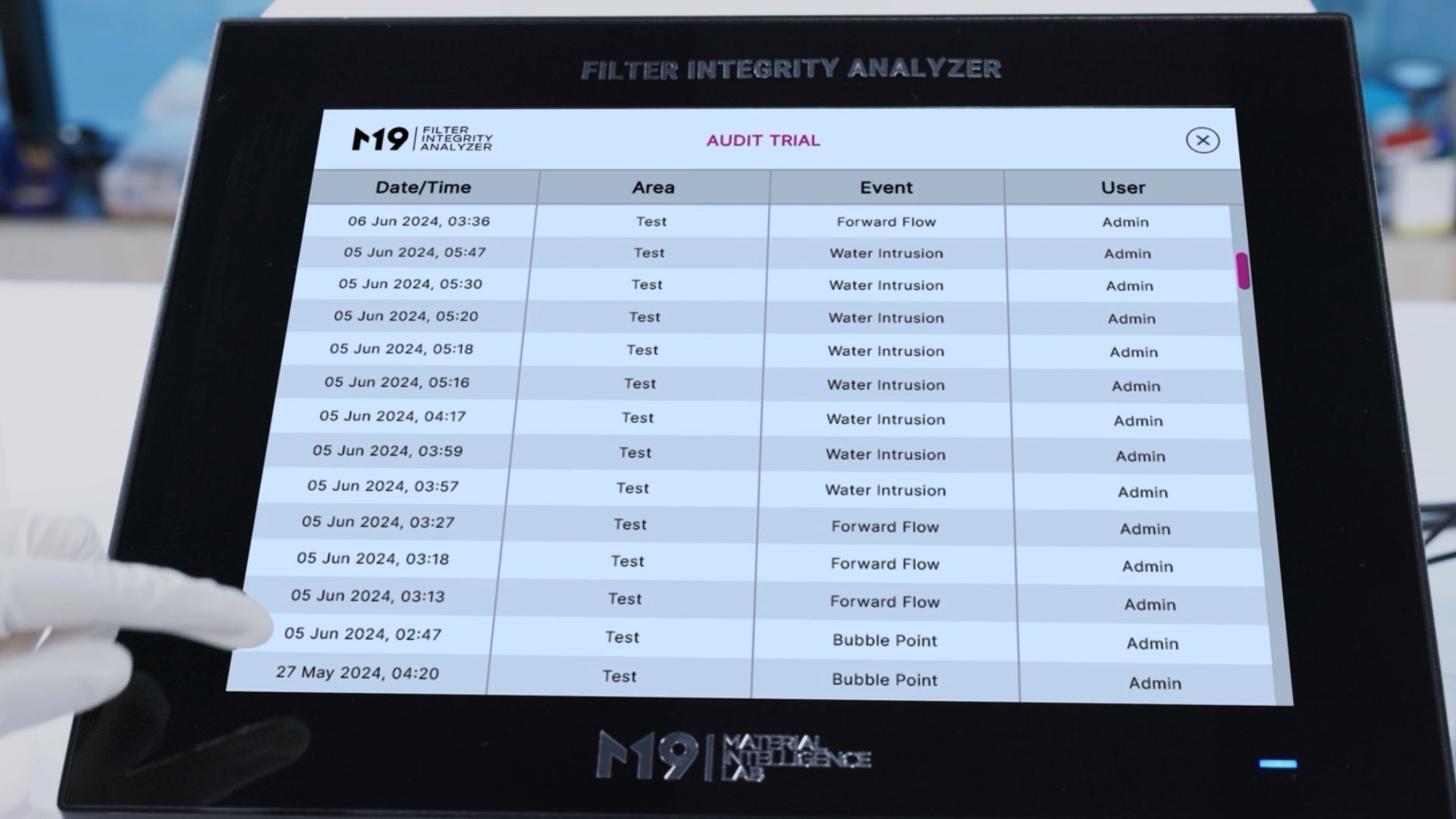
- Full 21 CFR Part 11 validation with comprehensive audit trail
- ARM7 processor delivers analysis 80% faster with 99.999% accuracy
- We are Trusted and Certified by:




The M19 Filter Integrity Analyzer is a precise, easy-to-use system that delivers confidence in every test. It verifies sterilizing and process filters with standardized methods, real-time control, and audit-ready data for regulated environments.
FILTER INTEGRITY ANALYZER
- 21 CFR Part 11 Compliant
- 5x Faster
- 2-Year Extended Warranty
MULTIPLE TESTS
- Bubble Point Test
- Forward Flow Test
- Water Intrusion Test
- Pressure Decay Test
- Combined Forward Flow
+ Bubble Point Test - Automated pre & post-use checks

- 15" Color LCD Touch Screen
- Unlimited Data Storage
- AI-enabled with LAN connectivity
- Integrated testing methods
- Bubble Point: 50-8000 ± 50 mbar
- Forward Flow: 0.1-1000 ± 0.1 ml/min
- Water Intrusion: 0.01-100 ± 0.01 ml/min
- Pressure Decay: 50-8000 ± 1 mbar
- Built in thermal printer or network/PDF exports

- 5x Faster results
- 21 CFR Part 11 compliant with audit trail
- Multiple user access levels (Operator, Supervisor, Administrator)
- Fast installation, M19 Expert training, and intelligent method setup
- IQ/OQ documentation and M19 Expert support for seamless validation
- 2-year warranty with spare parts assurance
 |
 |
 |
 |
 |
 |
 |
 |
Our engineers anticipate and resolve issues quickly, coordinate with your team end-to-end,
and keep validation activities and daily operations reliably on track.

Trusted globally by top pharma and research labs, the M19 Filter Integrity Analyzer delivers uncompromising accuracy for critical steps, preserves sterility, and provides the documented results regulators expect.










Connect With Our Industry ExpertsGET A FREE ONSITE/ONLINE DEMO
Ready to move forward? Connect with our experts for a personalized demo, quote, or consultation. We guarantee a response within 24 hours. |







